In recent years a variety of ranking systems have been developed by which species can be prioritized for the purpose of conservation. The nonprofit environmental organization NatureServe, for example, prioritizes the worldwide biota globally (G-ranks), nationally (N-ranks) or regionally (S-ranks) into five categories: 1 = critically imperiled, 2 = imperiled, 3 = vulnerable, 4 = apparently secure, and 5 = secure. The International Union for the Conservation of Nature also uses five ranks: CR = critically endangered, EN = endangered, VU = vulnerable, NT = near threatened and LC = least concern. A system of four ranks was advocated in the spurious review of Johnson et al. (2013), E = endangered, T = threatened, V = vulnerable, and CS = currently stable. A review of the many methods by which species have been categorized according to perceived conservation concern has been offered by Munton (1987).
Although the appeal of such systems to the natural resource agencies charged with protection of potentially endangered species is undeniable, such concepts as "threat" or "peril" or "endangerment" are by their nature entirely subjective. And although some connection is almost certainly made between conservation status rank and rarity in the minds of natural resource managers, the relationship has never been formally explored, at least in the case of freshwater snails. So, in 2011 we first suggested a ranking system based on incidence, offered as a compliment to (if not necessarily as an objective substitute for) the subjective systems of conservation status ranking currently in vogue.
> Version history
FWGNA synthesis v1.0 was initially proposed and forcefully advocated in essays posted on the FWGNA blogs of 12Dec11, 9Jan12, and 19Mar12. It was published in March 2012 as "FWGNA Circular No. 1" [pdf]. The database at that juncture contained 8,864 records of 57 species and subspecies, covering southern Atlantic drainages only. The distribution of commonness and rarity for those 57 species seemed to fit a lognormal model. Thus, a parametric system of incidence rankings was suggested, the rarest 5% being ranked F1, the remainder being divided by standard deviations around the mean: F2, F3, F4, and F5.
FWGNA synthesis v2.0 was proposed in the FWGNA website expansion of October 2013 and developed in the FWGNA blog post of 9Dec13. The analysis at that juncture was of 11,471 records collected from the Atlantic drainages of Georgia to the New York line. Number-theoretical analysis (EstimateS, Colwell 2013) returned no evidence that any rare species might have been missed in the list of 67 species (combining subspecies) returned.
The distribution of commonness and rarity demonstrated by the 67-species fauna did not appear lognormally distributed, but rather bimodal, with a secondary peak of high-incidence species. Thus, we shifted to a nonparametric model inspired by the work of Gaston (1994), suggesting a quartile system of I-ranks (I for incidence ), again setting aside the rarest 5% as I-1. We also introduced the concept of peripheral species at that time, with the related phenomena of pseudo-rarity and non-apparent rarity.
FWGNA synthesis v2.1 was announced in my blog post of 19Nov15 and went up on the FWGNA website that same day. The analytical methods remained identical to that of version 2.0, although the database was expanded to 12,211 records and 69 species. This was the version published in Volume 1 of the FWGNA series by Dillon et al (2019).
FWGNA synthesis v3.0. All versions of the FWGNA synthesis from 2012 2019 focused exclusively on the fauna of the Atlantic drainages. On 19June19 we expanded our analysis to include both the fauna of the Tennessee drainage above the Alabama line and the fauna of The Ohio above the mouth of the Tennessee/Cumberland, a total of 19,643 records. We tallied 102 freshwater gastropod species in that expanded region (lumping 9 additional subspecies within), set aside 6 (mostly stygobiontic) species as being inadequately sampled to rank, and ranked the remaining 96 by quartiles.
FWGNA synthesis 3.1. On 12May22 we announced the release of FWGNA Synthesis v3.1, an incremental (but not negligible) increase in our coverage beyond East Tennessee to include the entire freshwater gastropod fauna of the Tennessee/Cumberland drainage system, including North Alabama, Southern Kentucky, Middle Tennessee, and chips from the corners of Georgia and Mississippi. The FWGTN database increased from 1,798 records to 4,003. We also added 117 records to our FWGO database, collected since 2019, and 79 records to our older Atlantic databases, bringing the total records analyzed to 22,044. This was the version published in Volume 5 of the FWGNA series by Dillon and colleagues (2023).
> Version 3.2
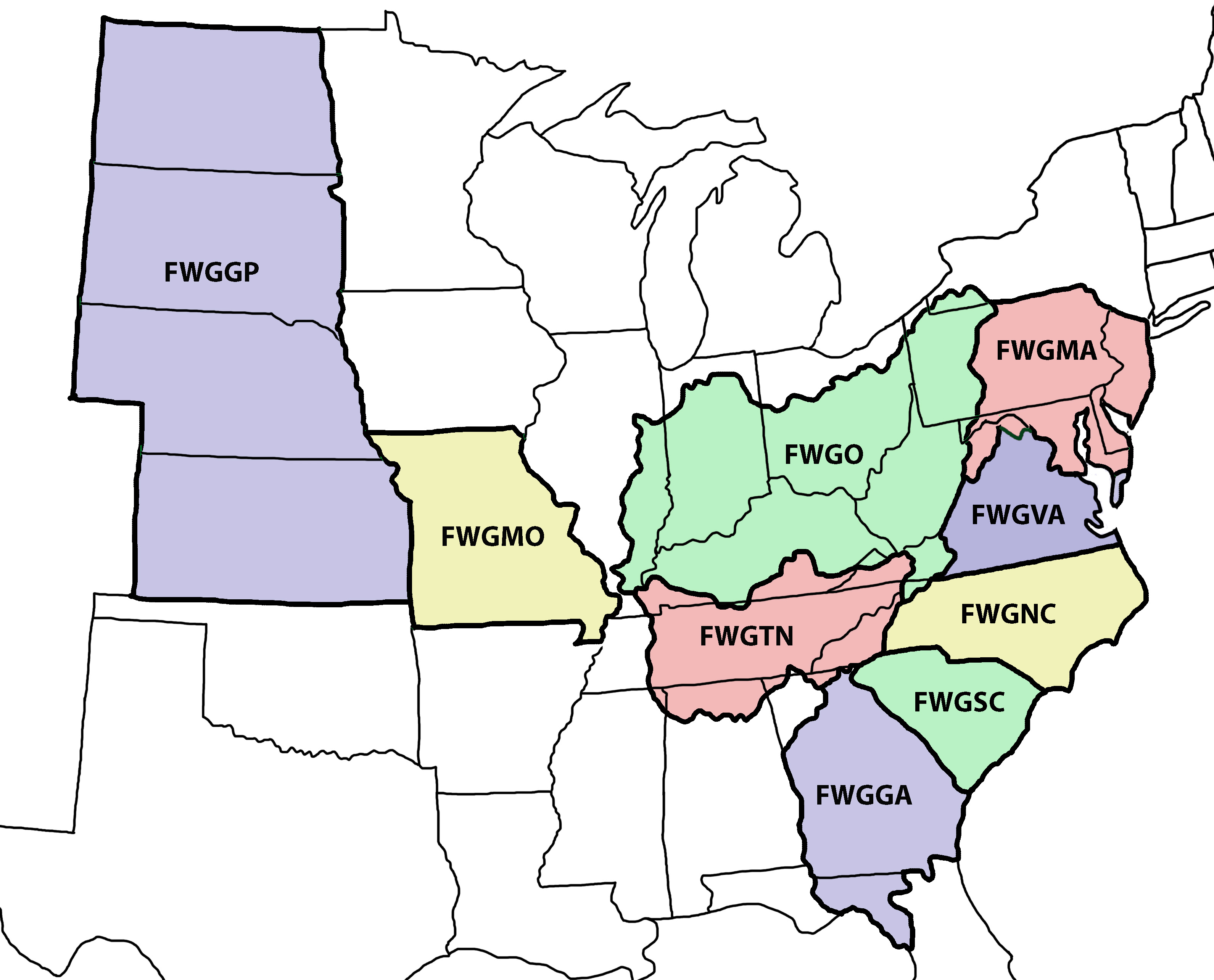
In June of 2026 we released another incremental-but-not-negligible expansion of our coverage to include Missouri (1,760 records) and the four states of the Great Plains (1,482 records). We also added the Gulf drainages of Georgia on that date, expanding into the panhandle of Florida, raising the FWGGA portion of the database from 960 to 1,610 records. Smaller increases were logged in the Carolinas (5,815), Virginia and the MidAtlantic States (5,469), the Tennessee/Cumberland (4,019) and the Ohio drainages (5,377). The total database analyzed in June 2026 increased from 22,044 records to 25,468.
We identified a total of 127 species of freshwater gastropods, with 27 subspecies, in that vast study area, touching all or part of 23 states. The incidence of these 154 species and subspecies, that is, the number of discrete sites at which each was observed, is tabulated by region: Southeast (7,370 records), MidAtlantic (5,460), Tennessee/Ohio (9,396), and The West (3,242) as a simple excel spreadsheet downloadable here [Synthesis32.xls].
The ecological requirements of this fauna are exceptionally diverse. Fourteen of the species are stygobionts obligately adapted to caves and other subterranean environments. These are all hydrobioid taxa: the amnicolids A. stygia and Antroselates, the cochliopid Antrobia, the lithoglyphid Holsingeria, and ten species of Fontigens. It seems unlikely to us that the incidences tabulated in Synthesis32.xls accurately reflect the abundances of these 14 species.
Ecologically, Pomatiopsis lapidaria is a land snail. Essentially all 103 P. lapidaria records in our database have come from museums, primarily in collections from forest litter. As was the case with the 14 stygobiontic species, it seems likely to us that our sampling methodology significantly underestimates the true abundance of P. lapidaria. It seems advisable therefore to exclude these 15 species from further analysis, subtracting 166 records.
The FWGNA Project accepts the standard definition of the word subspecies under the Modern Synthesis: populations of the same species in different geographic locations, with one or more distinguishing traits (Dillon 2019). Since under our definition the heritability of the morphological traits forming the basis of a subspecific distinction cannot be assumed, we lumped the 27 subspecific nomina under their parent species for further analysis, reducing the species count to 154 15 27 = 112.
In Table Synthesis32.pdf we have ordered the adequately-sampled 112 full species of freshwater gastropods inhabiting our 23-state study area by their overall incidence in the FWGNA database. Unsurprisingly, the most common species would appear to be Physa acuta, with 4,258 records. Physa acuta is a cosmopolitan invasive, native to North America but spread to five other continents (Dillon et al. 2002) and was the most common species in all four of the regions as well as in the total. The 1,568 records of the next-most common species, Campeloma decisum, amount to less than half of the P. acuta records. Interestingly, Campeloma retained its second-place rank only in the Southeastern drainages. In the other regions, tempered pride of second place went to Leptoxis carinata in the MidAtlantic drainages, Ferrissia rivularis in the Tennessee/Ohio, and Helisoma trivolvis in The West.
In striking contrast, five species were represented in the freshwater gastropod fauna of our 23-state study area by but a single record, even setting aside the 14 species we consider undersampled. Another three species were represented by just two records, and another six species by just three.
> Rarity
Rabinowitz (1981) has famously pointed out that there are seven forms of rarity. We have no data on population sizes, however, nor any rigorous measure of habitat specificity for our 112 freshwater gastropod species. Our analysis here has focused entirely upon geographical distributions. The species found in the most spots are common by our definition, and those in the fewest spots are rare. This may be termed incidence rarity.
Convention would dictate that some special consideration might be extended to the extreme 5% of any distribution, normally distributed or otherwise. Thus, we suggest that the 5% of the species demonstrating the lowest number of incidences in a biota under consideration be assigned the rank I-1, by analogy to the G1 rank of NatureServe. The I prefix here designates incidence, to emphasize that the present ranking system is based on incidence data, rather than subjective impressions of global imperilment.
Gaston (1994) has offered an admirable review of the term rare in all its various origins and biological usages. On the basis of clarity, versatility, consistency, and ease of use, he has suggested that the term "rare" be defined as the first quartile of the frequency distribution of species abundances. So because we have just set aside the 5% of species with the lowest incidence frequency as I-1, perhaps the 20% of the species remaining in the lowest quartile ought to be designated I-2. Then a straightforward application of Gaston s system would suggest that the second quartile (between the rarest set and the median) be designated I-3, and the third quartile I-4, and the fourth (most common) quartile designated I-5.
Then setting aside the terrestrial Pomatiopsis lapidaria and the 14 stygobiontic species, the rightmost ( FWGNA ) column of Table Synthesis32.pdf shows the vector of I-ranks resulting from an application of Gaston s system to the 127 - 15 = 112 freshwater gastropod species inhabiting our 23-state study area.
> Peripheral Species
Murray et al. (1999) reported that a remarkable 91% of the species in the tail of a rank-abundance curve generated from the canopy-forming vegetation of the Australian dry sclerophyll woodland were significantly more abundant elsewhere. Murray and colleagues called these 91% the somewhere-abundant species, to distinguish that group from the 9% that were everywhere-sparse.
Similar phenomena have been noted in many animal communities. Gaston (1994) has reported that the vast majority of the British bird species demonstrating incidence rarity are vagrants, which he defines as not permanent members of the assemblage, do not breed, or do not have self-sustaining populations. Gaston notes that other terms that have been applied to describe such species include accidentals, casuals, immigrants, incidentals, strays, tourists and tramps. Magurran & Henderson (2003) have added the term occasionals for rare species in estuarine fish communities. The literature of plant community ecology includes the terms "peripheral" and "waif."
Similar to the situation described by Murray and colleagues, the data in Table Synthesis32.pdf suggest to us two categories of rare species, the somewhere-abundant and the everywhere-sparse. But we are not aware of any term in malacology analogous to vagrant or occasional, probably because such terms imply greater dispersal capability than is ordinarily assumed for freshwater mollusks.
We therefore suggest adopting the botanical term peripheral for use in mollusk community ecology to describe the situation where a rare species is somewhere-else-abundant. We formally define a peripheral species as demonstrating less than median incidence in a region under study, but greater than median incidence elsewhere. And we suggest that all non-peripheral species in a study region be called core species.
Although there are few rigorous estimates of the relative incidence of freshwater gastropod species outside the present analysis, our reading of the malacological literature suggests to us that 25 of the 56 species listed below the median in Table Synthesis32.pdf probably demonstrate above-median incidence elsewhere. Five of these are exotic invasives Bithynia tentaculata, Potamopyrgus antipodarium, Pyrgophorus parvulus, Melanoides tuberculata and Pomacea maculata. The excellent New York survey of Jokinen (1992) and the Canadian survey of Clarke (1981) suggest to us that the following 10 species are more common to the north of our study area: Marstonia lustrica, Probythinella emarginata, Lymnaea catascopium, L. stagnalis, Helisoma campanulatum, Gyraulus deflectus, G. circumstriatus, G. crista, Physa jennessi, and Aplexa elongata.
The Florida survey of Thompson (1999) suggests that the following 7 species are more common further south: Neritina usnea, Pomacea paludosa, Aphaostracon monas, Pleurocera curvicostata, Floridobia floridana, Biomphalaria havanensis, and Hebetancylus excentricus. Our reading of the continental literature suggests that Promenetus umbilicatellus and the poorly-known Lymnaea cockerelli are more common in the West, and that the poorly-understood Pleurocera modesta is more common in upper Coosa drainages draining south through Georgia into the Mobile Basin. A lower-case p has been appended to the incidence ranks of all 25 of these species in Table Synthesis32.pdf, to indicate their (hypothesized) peripheral status in our 23-state study region.
> Pseudo-rarity and Non-apparent rarity
The 6 core species marked I-1 and the 12 core species marked I-2 in the rightmost column of Table Synthesis32.pdf are clearly rare, demonstrating incidences in the bottom quartile. But Gaston (1994) has suggested a new term to describe the 28 18 = 10 peripheral species occupying positions in the bottom quartile of Table Synthesis32.pdf, pseudorare. This is because the evidence suggests that they are not rare elsewhere.
Indeed, those 10 pseudo-rare species have displaced 10 species that should have occupied their lines in the bottom quartile. Thus the 10 core species marked I-3* in the rightmost column of Table Synthesis32.pdf (without the modifier "p") all demonstrate non-apparent rarity. The evidence suggests that they are legitimately rare, and deserved rank in the bottom quartile, but their rarity was obscured by the 10 pseudo-rare, peripheral species.
> Future Prospects
Note that seven of the species listed in Table Synthesis32.pdf are exotic: the five listed four paragraphs above plus Cipangopaludina japonica and C. chinensis, populations of which have now risen to I-3 commonness in our study area. We have elected to treat these seven no different from native North American species.
Indeed, at least eight of our native species have been considered invasive elsewhere: Physa acuta, Helisoma trivolvis, H. scalare, Lymnaea columella, Hebetancylus excentricus, Ferrissia fragilis, Viviparus georgianus and V. subpurpureus, and seem to have expanded their ranges here in North America in modern times. And at least two other species may have obtained representation in Table Synthesis32 by artificial introduction (Pomacea paludosa and Biomphalaria havanensis), although demonstrating little potential to spread.
We do not expect the designations in the rightmost column of Table Synthesis32.pdf to remain static. Indeed, we expect the opposite. Currently FWGNA surveys are expanding south into the Florida peninsula, North into New York and west through Arkansas. The I-ranks currently shown in Table Synthesis32.pdf should be considered subject to revision for quite a few years to come.
> Essays
- Our initial effort to develop a (parametric) theory of commonness and rarity for freshwater gastropods was based on the incidence of 57 species in four states only. This analysis was introduced and justified in my blog posts of 12Dec11 and 9Jan12: "Toward the Scientific Ranking of Conservation Status."
- I introduced the first nine-state, nonparametric version of this analysis in my blog post of 9Dec13, "What is Rarity?" That particular essay focused on Gaston's quartile definition. The 2013 FWGNA database at that juncture contained 11,471 records from the Atlantic drainages, representing 67 species.
- I focused on the subjects of "peripheral" species, pseudo-rarity and non-apparent rarity in my follow-up essay of 6Jan14, "Why is Rarity?"
- Version 2.1 of the FWGNA synthesis (12,211 records, 69 species) was an incremental (although not negligible) expansion of our 2013 analysis. It was announced in my blog post of 19Nov15.
- I announced Version 3.0 of the FWGNA synthesis on 19June19, expanding coverage west over the Blue Ridge to include drainages of The Ohio and East Tennessee, bringing the total records to 19,643 and the total species to 102.
> References
Clarke, A. H. 1981. The Freshwater Molluscs of Canada. National Museum of Natural Sciences, National Museums of Canada, Ottawa.
Colewell, R. K. 2013. EstimateS: Statistical estimation of species richness and shared species from samples. Version 9. User's Guide and application published at: http://purl.oclc.org/estimates.
Dillon, R.T., Jr. (2019) What is a subspecies? Pp 77 - 82 in The Freshwater Gastropods of North America Volume 3, Essays on the Prosobranchs. FWGNA Press, Charleston. [4Feb14]
Dillon, R.T., Jr., M.J. Ashton, W.K. Reeves, T.P. Smith, T.W. Stewart, & B.T. Watson (2019) Atlantic drainages, Georgia through Pennsylvania. Freshwater Gastropods of North America, Volume 1. FWGNA Press. 199 pp. [html]
Dillon, R.T. Jr., M. Kohl, R. Winters, M. Pyron, W.K. Reeves, G.T. Watters, K. Cummings, J. Bailey, & M. Whitman (2023) Ohio, Cumberland, and Tennessee River Systems. Freshwater Gastropods of North America, Volume 5. FWGNA Press, Charleston, SC. 315 pp. [html]
Dillon, R. T., A. R. Wethington, J. M. Rhett and T. P. Smith. (2002) Populations of the European freshwater pulmonate Physa acuta are not reproductively isolated from American Physa heterostropha or Physa integra. Invertebrate Biology 121: 226-234. [PDF]
Gaston, K. J. 1994. Rarity. Chapman & Hall, London. 205 pp.
Johnson, P.D. Bogan, Brown, Burkhead, Cordeiro, Garner, Hartfield, Lepitzki, Mackie, Pip, Tarpley, Tiemann, Whelan & Strong 2013. Conservation status of freshwater gastropods of Canada and the United States. Fisheries 38: 247- 282.
Jokinen, E. H. 1992. The Freshwater Snails (Mollusca: Gastropoda) of New York State. Albany: New York State Museum. 112 pp.
Munton, P. 1987. Concepts of threat to the survival of species used in Red Data books and similar compilations. Pp 72- 95 In The Road to Extinction (R. Fitter & M. Fitter, eds.) IUCN/UNEP. Gland, Switzerland
Magurran, A. E. & P. A. Henderson 2003. Explaining the excess of rare species in natural species abundance distributions. Nature 422: 714-716.
Murray, B. R., B. L. Rice, D. A. Keith, P. J. Myerscough, J. Howell, A. G. Floyd, K. Mills & M. Westoby 1999. Species in the tail of rank-abundance curves. Ecology 80: 1806-1816.
Rabinowitz, D. 1981. Seven forms of rarity. Pp 205 217 in The Biological Aspects of Rare Plant Conservation (H. Synge, ed.) Wiley, NY.
Thompson, F. G. 1999. An identification manual for the freshwater snails of Florida. Walkerana 10 (23): 1 96.